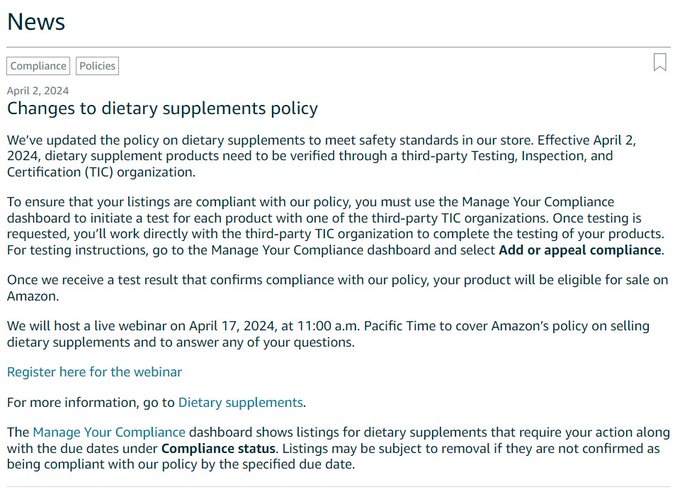
This new set of rules includes the requirement for Content Claims Verification.
This is a HUGE deal for the whole industry. It will likely add up to 100s of millions of dollars in testing per year.
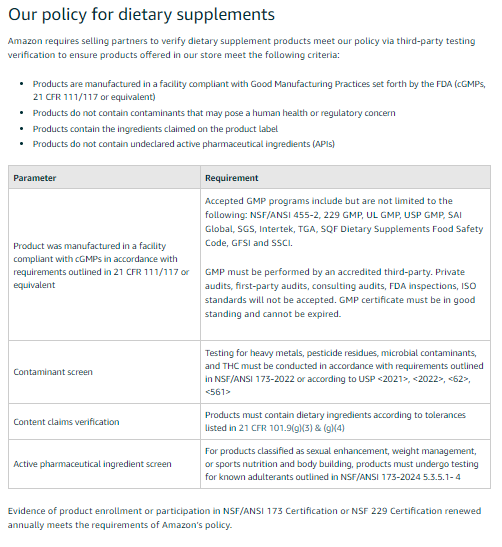
NSF International will now become the official third party certifier for Dietary Supplements.
Unclear who is paying for these tests and certifications. Amazon or the Seller?
@amznsellerhelp
do you know?
Content verification tests are EXPENSIVE and depending on the number of ingredients in a product, could be cost-prohibitive for certain supplements.
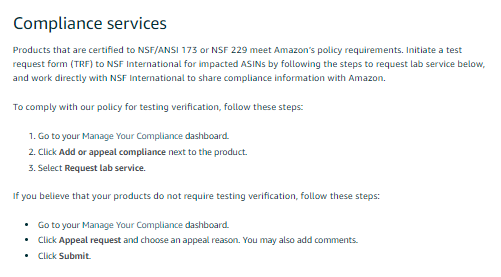
This is going to be VERY interesting.
Dietary Supplements is an industry with a ton of nuance. So very curious as to how Amazon will navigate this.
Examples of this:
Different testing methods can result in differing results.
Some labs don't know how to test certain ingredients.
A single bottle can test differently than the majority of a lot.
How will Amazon handle retesting (which is common)?
How will Amazon handle chain of custody of the product? E.g. What if it accidently sends a returned product for testing?
What is the path for a product that tests at 99% of content claims? Is it just suspended immediately?
@amznsellerhelp
I am sure there will be many questions in the comments.
Here is the News post on Amazon